Chinese Journal of Tissue Engineering Research ›› 2015, Vol. 19 ›› Issue (7): 1077-1081.doi: 10.3969/j.issn.2095-4344.2015.07.017
Previous Articles Next Articles
Role of estrogen in bone metabolism
Li Hong-liang, Si Jia-wen, Shen Guo-fang
- Department of Oral and Craniomaxillofacial Science, Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai Key Laboratory of Stomatology, Shanghai 200011, China
-
Online:2015-02-12Published:2015-02-12 -
Contact:Shen Guo-fang, M.D., Chief physician, Department of Oral and Craniomaxillofacial Science, Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai Key Laboratory of Stomatology, Shanghai 200011, China -
About author:Li Hong-liang, Studying for master’s degree, Department of Oral and Craniomaxillofacial Science, Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai Key Laboratory of Stomatology, Shanghai 200011, China -
Supported by:the National Natural Science Foundation of China, No. 81371122
CLC Number:
Cite this article
Li Hong-liang, Si Jia-wen, Shen Guo-fang. Role of estrogen in bone metabolism[J]. Chinese Journal of Tissue Engineering Research, 2015, 19(7): 1077-1081.
share this article
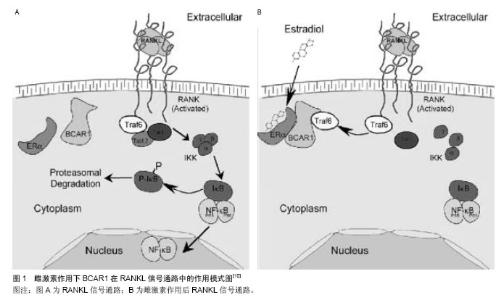
2.1 雌激素对破骨细胞的作用 Oursler 于1991年证实了破骨细胞中雌激素受体的存在[6],说明雌激素对破骨细胞有直接的调控作用。Martin-Millan发现特异性敲除破骨细胞雌激素受体α导致了松质骨的损失[7],这是由于减少了破骨细胞的凋亡,使破骨细胞的寿命的延长。 此外在促使破骨细胞凋亡的过程中,雌激素阻断了核因子κB受体活化因子配体/巨噬细胞集落刺激因子介导的转录过程[8],通过降低c-jun的活性,抑制核因子κB受体活化因子配体诱导的破骨细胞的分化[9]。雌激素通过诱导雌激素受体α结合于BCAR1蛋白上,来抑制核因子ΚB受体活化因子配体刺激的单核细胞向破骨的分化,隔断了TRAF6,导致了核因子κB活性的降低,阻断了核因子ΚB受体活化因子配体诱导的破骨现象,见图1[10]。除了直接对破骨细胞发生作用,雌激素还间接调节破骨细胞的形成及活性。体内体外实验均表明雌激素亦通过对成骨向细胞、T细胞、B细胞抑制核因子ΚB受体活化因子配体的生成,同时增加核因子ΚB受体活化因子配体的抑制物-骨保护素的生成[11]。 在小鼠动物模型中,雌激素被证实调节一系列骨吸收相关因子,包括白细胞介素1、白细胞介素6、肿瘤坏死因子α、巨噬细胞集落刺激因子、前列腺素[12-17],另外,肿瘤坏死因子α的促吸收作用可以被可溶性的一型肿瘤坏死因子受体逆转[18]。在绝经后雌激素显著下降的妇女中应用阿那白滞素及依那西普特异性阻碍白细胞介素1和肿瘤坏死因子α,其骨吸收相关标志物不再上调,和小鼠模型中一致,这些免疫细胞因子的调节作用在人类中也很重要[19]。 有学者提出伴随着雌激素水平的显著下调而出现的快速的骨丧失,是由于T细胞的扩增到时骨微环境中的肿瘤坏死因子α水平上升引起的,同时间接地促进了破骨细胞的形成[20]。在缺乏T淋巴细胞的裸小鼠中并未出现因卵巢切除而导致的骨丢失也证实了这一点[21]。然而其他研究者在裸大鼠、RAG2和TCR-α缺陷小鼠(均缺乏有功能的T淋巴细胞)进行卵巢切除后发现,这些老鼠均出现了与野生老鼠类似的骨丢失的现象[22-23]。 总之,雌激素通过直接或间接的作用对破骨细胞的形成、活性、寿命产生了影响。一方面,雌激素通过雌激素受体α对破骨细胞直接的调控作用或许占据了主导,另一方面雌激素通过T细胞和成骨细胞产生的对破骨细胞间接的调控作用亦为重要。 2.2 雌激素对成骨细胞的作用 1988年Komm等[24]发现成骨细胞中有雌激素受体(estrogen receptors,ERs)存在,提示雌激素对成骨细胞有调节作用。随着成骨前体细胞和骨细胞上雌激素受体的发现,越来越多的研究证明雌激素对成骨过程具有直接作用,也具有不依赖雌激素受体的间接的作用。 2.2.1 雌激素参与调节成骨细胞增殖与分化 成骨细胞的增殖与凋亡对维持骨代谢平衡起重要作用[25-26],雌激素对成骨细胞表现为抗凋亡与促进分化的作用。 Kousteni等[27]研究发现雌激素能够抑制成骨细胞的凋亡,延长其寿命,从而增加了每个成骨细胞的成骨能力。其作用是通过雌激素受体介导的调节激酶的快速激活实现的[28]。有报道称成骨细胞的凋亡与B淋巴细胞瘤2基因有关,雌激素能够通过促进B淋巴细胞瘤2基因的转录与表达[29],阻断糖皮质激素所引起的成骨细胞的凋亡[30],亦有人报道雌激素通过上调热休克蛋白而减少肿瘤坏死因子对成骨细胞的凋亡作用[31]。雌激素减少成骨细胞的凋亡也可通过Src/Shc/ERK信号通路的激活以及JNK的下调,改变了一系列转录因子的活性包括Elk-1、cJun/cFos等来实现[28]。 雌激素对成骨细胞的增殖、成骨标志物(Ⅰ型胶原、骨钙素、碱性磷酸酶)的表达及细胞因子等有直接的作用[32],对小鼠的骨髓进行培养发现,雌激素能够促进成骨细胞的分化,通过雌激素受体的作用,促进钙盐的沉积于矿化[33-35]。雌激素对成骨细胞的作用主要通过雌激素受体机制[36]。雌激素受体分为两种,雌激素受体α与雌激素受体β[37-39],两者虽然再结构上有高度的同源性,但功能存在争议。研究证明,两者在雌激素调节成骨细胞的过程中发挥了不同的作用。在小鼠的成骨细胞系中敲除雌激素受体α后发现其几乎没有成骨的表型,在敲除雌激素受体α后的小鼠中发现皮质骨有明显的减少,然而松质骨并未受影响[40]。亦有学者称雌激素受体α缺乏抑制了鼠骨骼的纵向生长,而雌激素受体β缺乏了促进纵向的生长,表明在纵向上两者起着不同的作用,然而在对骨小梁密度方面的影响是相同的[41]。关于两种受体的不同作用仍有待进一步研究。 2.2.2 雌激素参与调节成骨细胞核因子κB活性 有体内实验表明:雌激素的缺乏使得成骨细胞中的核因子κB活性显著增加[42],另外,在去卵巢大鼠中抑制核因子κB活性可以减少骨吸收和骨形成的失衡,从而减少骨丢失的发生。核因子κB的抑制导致了Fos相关抗原1(Fra-1)表达的增加,Fra-1是骨基质形成过程中的重要的转录因子。在体外诱导成骨细胞系分化时,通过加入核因子κB的抑制剂IKK-DN和SR-IκBα,能够促进细胞的成骨向分化[42]。进一步研究证实,尽管雌激素对核因子κB没有直接的调控作用,但是它能够显著地降低肿瘤坏死因子α介导的核因子κB活性[43],从而激活了转化生长因子β介导的Smad信号通路[44],促进了MC3T3成骨细胞系以及骨髓间质干细胞分化与矿化的功能。另外,也有报道称核因子κB通过影响Smad7信号通路而作用于Saos2骨肉瘤细胞而抑制其成骨分化[45]。然而雌激素对核因子κB活性的具体作用机制尚不完善,仍需进一步研究证实。 2.2.3 雌激素参与调节成骨细胞氧化应激反应 活性氧的水平对细胞的许多功能都会产生影响,包括细胞凋亡的时间,老化,以及年龄有关的相关疾病[46]。体内过多的积累活性氧会引起许多组织的改变,其中也包含骨组织[47]。 一系列的研究表明,在年长小鼠的骨组织内,氧化应激的标志物的表达较高[45]。氧化应激的产物活性氧,能够减弱成骨细胞的生成,并减少成骨细胞和骨细胞的寿命,相反,活性氧是破骨细胞生成、发挥功能所必须的。氧化应激抑制了骨形成,其中一部分是由于阻断了Wnt信号通路以及将β-catenin从TCF转移至FoxO介导的转录因子结合位点[48],同时雌激素缺乏对骨的影响可以通过抗氧化剂来抵消[45]。总的来说,年龄因素和雌激素缺乏对骨形成的作用可能是通过氧化应激反应来实现的。"
| [1]Jonasson G, Kiliaridis S,Gunnarsson R.Cervical thickness of the mandibular alveolar process and skeletal bone mineral density.Acta Odontol Scand.1999;57:155-161. [2]Taguchi A, Suei Y, Ohtsuka M, et al.Relationship between bone mineral density and tooth loss in elderly Japanese women.Dentomaxillofac Radiol. 1999;28:219-223. [3]Becker W, Hujoel PP, Becker BE, et al. Osteoporosis and implant failure: an exploratory case-control study.J Periodontol. 2000;71:625-631. [4]Klemetti E, Vainio P,Kroger H. Craniomandibular disorders and skeletal mineral status.Cranio.1995;13:89-92. [5][No authors listed].Zoledronic acid: new indication. No therapeutic advantage in postmenopausal osteoporosis. Prescrire Int. 2008;17:143. [6]Oursler MJ, Osdoby P, Pyfferoen J, et al. Avian osteoclasts as estrogen target cells.Proc Natl Acad Sci USA. 1991;88:6613-7. [7]Martin-Millan M, Almeida M,Ambrogini E, et al.The estrogen receptor-alpha in osteoclasts mediates the protective effects of estrogens on cancellous but not cortical bone.Mol Endocrinol. 2010;24:323-334. [8]Tera Tde M, Prado RF, De Marco AC, et al.The RANK/ RANKL/ OPG interaction in the repair of autogenous bone grafts in female rats with estrogen deficiency. Braz Oral Res. 2014 Jan-Feb;28(1). pii: S1806-83242014000100261. [9]Srivastava S,Toraldo G,Weitzmann MN,et al.Estrogen decreases osteoclast formation by down-regulating receptor activator of NF-kappa B ligand (RANKL)-induced JNK activation. J Biol Chem.2001;276:8836-8340. [10]Robinson LJ,Yaroslavskiy BB,Griswold RD,et al.Estrogen inhibits RANKL-stimulated osteoclastic differentiation of human monocytes through estrogen and RANKL-regulated interaction of estrogen receptor-alpha with BCAR1 and Traf6.Exp Cell Res.2009; 315: 1287-301. [11]Hofbauer LC, Khosla S, Dunstan CR, et al. Estrogen stimulates gene expression and protein production of osteoprotegerin in human osteoblastic cells.Endocrinology. 1999;140:4367-4370. [12]Ammann P, Rizzoli R, Bonjour JP, et al. Transgenic mice expressing soluble tumor necrosis factor-receptor are protected against bone loss caused by estrogen deficiency.J Clin Invest.1997; 99: 1699-1703. [13]Kimble RB, Srivastava S, Ross FP, et al. Estrogen deficiency increases the ability of stromal cells to support murine osteoclastogenesis via an interleukin-1and tumor necrosis factor-mediated stimulation of macrophage colony-stimulating factor production.J Biol Chem.1996;271:28890-28897. [14]Kimble RB, Vannice JL, Bloedow DC, et al. Interleukin-1 receptor antagonist decreases bone loss and bone resorption in ovariectomized rats.J Clin Invest. 1994;93:1959-1967. [15]Manolagas SC,Jilka RL. Bone marrow, cytokines, and bone remodeling. Emerging insights into the pathophysiology of osteoporosis.N Engl J Med. 1995;332:305-311. [16]Pacifici R, Brown C, Puscheck E, et al.Effect of surgical menopause and estrogen replacement on cytokine release from human blood mononuclear cells.Proc Natl Acad Sci USA. 1991;88:5134-5138. [17]Tanaka S, Takahashi N, Udagawa N, et al. Macrophage colony-stimulating factor is indispensable for both proliferation and differentiation of osteoclast progenitors.J Clin Invest. 1993; 91:257-263. [18]Kitazawa R, Kimble RB, Vannice JL, et al.Interleukin-1 receptor antagonist and tumor necrosis factor binding protein decrease osteoclast formation and bone resorption in ovariectomized mice.J Clin Invest. 1994;94:2397-2406. [19]Charatcharoenwitthaya N, Khosla S, Atkinson EJ, et al. Effect of blockade of TNF-alpha and interleukin-1 action on bone resorption in early postmenopausal women.J Bone Miner Res. 2007;22:724-729. [20]Roggia C, Gao Y, Cenci S, et al.Up-regulation of TNF-producing T cells in the bone marrow: a key mechanism by which estrogen deficiency induces bone loss in vivo.Proc Natl Acad Sci U S A. 2001;98:13960-13965. [21]Cenci S, Weitzmann MN, Roggia C, et al. Estrogen deficiency induces bone loss by enhancing T-cell production of TNF-alpha.J Clin Invest.2000;106:1229-1237. [22]Lee SK, Kadono Y, Okada F, et al. T lymphocyte-deficient mice lose trabecular bone mass with ovariectomy.J Bone Miner Res. 2006;21:1704-1712. [23]Sass DA,Liss T,Bowman AR,et al.The role of the T-lymphocyte in estrogen deficiency osteopenia.J Bone Miner Res. 1997;12: 479-486. [24]Komm BS, Terpening CM, Benz DJ, et al.Estrogen binding, receptor mRNA, and biologic response in osteoblast-like osteosarcoma cells.Science. 1988;241:81-84. [25]Gao B, Huang Q, Lin YS, et al.Dose-dependent effect of estrogen suppresses the osteo-adipogenic transdifferentiation of osteoblasts via canonical Wnt signaling pathway.PLoS One. 2014;9:e99137. [26]Qiu X, Jin X, Shao Z, et al.17beta-estradiol induces the proliferation of hematopoietic stem cells by promoting the osteogenic differentiation of mesenchymal stem cells.Tohoku J Exp Med.2014;233:141-148. [27]Kousteni S, Bellido T, Plotkin LI, et al.Nongenotropic, sex-nonspecific signaling through the estrogen or androgen receptors: dissociation from transcriptional activity.Cell. 2001; 104:719-730. [28]Kousteni S,Han L,Chen JR,et al.Kinase-mediated regulation of common transcription factors accounts for the bone-protective effects of sex steroids.J Clin Invest. 2003;111:1651-1664. [29]Pantschenko AG,Zhang W,Nahounou M,et al.Effect of osteoblast-targeted expression of bcl-2 in bone: differential response in male and female mice.J Bone Miner Res.2005; 20:1414-1429. [30]Gu G, Hentunen TA, Nars M, et al. Estrogen protects primary osteocytes against glucocorticoid-induced apoptosis. Apoptosis. 2005;10:583-595. [31]Cooper LF,Tiffee JC,Griffin JP,et al.Estrogen-induced resistance to osteoblast apoptosis is associated with increased hsp27 expression.J Cell Physiol. 2000;185:401-7. [32]Spelsberg TC, Subramaniam M, Riggs BL, et al. The actions and interactions of sex steroids and growth factors/cytokines on the skeleton.Mol Endocrinol. 1999;13:819-828. [33]Qu Q, Perala-Heape M, Kapanen A, et al. Estrogen enhances differentiation of osteoblasts in mouse bone marrow culture. Bone. 1998;22:201-209. [34]de Faria AN, Zancanela DC, Ramos AP, et al. Estrogen and phenol red free medium for osteoblast culture: study of the mineralization ability.Cytotechnology. 2015. [Epub ahead of print] [35]White J, Wilson G, Tucci MA, et al. The effects of sustained delivery of estrogen on bone strength and cardiovascular panels in osteoporotic female rats.Biomed Sci Instrum. 2014; 50:336-344. [36]Shapiro LF,Freeman K. The relationship between estrogen, estrogen receptors and periodontal disease in adult women.J Mich Dent Assoc. 2014;96:40-44. [37]Tang WR, Liu Y, Li LH, et al. [Fluid shear stress and raloxifene stimulates the proliferation of osteoblast through regulating the expresstion of beta-catenin and estrogen receptor alpha].Sichuan Da Xue Xue Bao Yi Xue Ban. 2014;45: 913-918. [38]Chow SK, Leung KS, Qin L, et al. Callus formation is related to the expression ratios of estrogen receptors-alpha and -beta in ovariectomy-induced osteoporotic fracture healing.Arch Orthop Trauma Surg.2014;134:1405-1416. [39]Henning P, Ohlsson C, Engdahl C, et al. The effect of estrogen on bone requires ERalpha in nonhematopoietic cells but is enhanced by ERalpha in hematopoietic cells.Am J Physiol Endocrinol Metab. 2014;307:E589-595. [40]Almeida MQ, Tsang KM, Cheadle C, et al. Protein kinase A regulates caspase-1 via Ets-1 in bone stromal cell-derived lesions: a link between cyclic AMP and pro-inflammatory pathways in osteoblast progenitors.Hum Mol Genet.2011;20: 165-175. [41]Lindberg MK, Alatalo SL, Halleen JM, et al. Estrogen receptor specificity in the regulation of the skeleton in female mice.J Endocrinol. 2001;171:229-236. [42]Chang J, Wang Z, Tang E, et al. Inhibition of osteoblastic bone formation by nuclear factor-kappaB.Nat Med. 2009;15: 682-689. [43]Yamaguchi M,Weitzmann MN. The estrogen 17beta-estradiol and phytoestrogen genistein mediate differential effects on osteoblastic NF-kappaB activity.Int J Mol Med. 2009;23: 297-301. [44]Li Y, Li A, Strait K, et al. Endogenous TNFalpha lowers maximum peak bone mass and inhibits osteoblastic Smad activation through NF-kappaB.J Bone Miner Res. 2007;22: 646-655. [45]Almeida M, Han L, Martin-Millan M, et al. Skeletal involution by age-associated oxidative stress and its acceleration by loss of sex steroids.J Biol Chem. 2007;282:27285-27297. [46]Eliseev RA, Schwarz EM, Zuscik MJ, et al. Smad7 mediates inhibition of Saos2 osteosarcoma cell differentiation by NFkappaB.Exp Cell Res. 2006;312:40-50. [47]Finkel T,Holbrook NJ.Oxidants, oxidative stress and the biology of ageing.Nature. 2000;408:239-247. [48]Manolagas SC,Almeida M. Gone with the Wnts: beta-catenin, T-cell factor, forkhead box O, and oxidative stress in age-dependent diseases of bone, lipid, and glucose metabolism. Mol Endocrinol. 2007;21:2605-2614. |
| [1] | Chen Jiming, Wu Xiaojing, Liu Tianfeng, Chen Haicong, Huang Chengshuo. Effects of silymarin on liver injury and bone metabolism induced by carbon tetrachloride in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1224-1228. |
| [2] | Zheng Xiaolong, He Xiaoming, Gong Shuidi, Pang Fengxiang, Yang Fan, He Wei, Liu Shaojun, Wei Qiushi. Bone turnover characteristics in patients with alcohol-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 657-661. |
| [3] | Yang Caihui, Liu Qicheng, Dong Ming, Wang Lina, Zuo Meina, Lu Ying, Niu Weidong. Serine/threonine protein kinases can promote bone destruction in mouse models of chronic periapical periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3654-3659. |
| [4] | Huo Hua, Cheng Yuting, Zhou Qian, Qi Yuhan, Wu Chao, Shi Qianhui, Yang Tongjing, Liao Jian, Hong Wei. Effects of drug coating on implant surface on the osseointegration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3558-3564. |
| [5] | Jiang Shengyuan, Li Dan, Jiang Jianhao, Shang-you Yang, Yang Shuye. Biological response of Co2+ to preosteoblasts during aseptic loosening of the prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(21): 3292-3299. |
| [6] | Liu Bo, Chen Xianghe, Yang Kang, Sun Changliang, Yu Huilin, Lu Pengcheng. Epigenetic reprogramming and exercise regulation of bone metabolism disorders [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3210-3218. |
| [7] | Huang Zhusong, Lin Yu, Chen Xiang, Lan Jinfu, Guan Yong, Gao Xi. Alcohol extract of Morinda officinalis improves lipid metabolism and bone metabolism in ovariectomized obese rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 205-210. |
| [8] | Wei Qin, Zhang Xue, Ma Lei, Li Zhiqiang, Shou Xi, Duan Mingjun, Wu Shuo, Jia Qiyu, Ma Chuang. Platelet-derived growth factor-BB induces the differentiation of rat bone marrow mesenchymal stem cells into osteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 2953-2957. |
| [9] | Guo Zhibin, Wu Chunfang, Liu Zihong, Zhang Yuying, Chi Bojing, Wang Bao, Ma Chao, Zhang Guobin, Tian Faming. Simvastatin stimulates osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 2963-2968. |
| [10] | Zhu Shiqiang, Xu Jianfeng, Hei Xiaoyan, Chen Yundong, Tian Xinbao, Zhang Jinchen, Lin Ruizhu. Effect of internal heat-type acupuncture needle therapy on the expression of type I collagen, matrix metalloproteinase-3 and osteopontin in the subchondral bone of rabbit knee osteoarthritis model [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2636-2642. |
| [11] | Zhou Yi, Chen Yueping, Zhang Xiaoyun, Lai Yu, Liao Jianzhao, Li Shibin. An exploration on mechanism of Shengyu Decoction in treating osteonecrosis of the femoral head based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2687-2696. |
| [12] | Chen Jiayun, Li Anan, Lü Zhaohui, Wu Zixuan, Cai Minjie, Huang Xuyan . Effect of long-term use of proton pump inhibitors on bone mineral density and bone metabolism: a Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2775-2780. |
| [13] | Wu Yukun, Han Jie, Wen Shuaibo. Mechanism of Runx2 gene in fracture healing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(14): 2274-2279. |
| [14] | Li Shengqiang, Xie Bingying, Chen Juan, Xie Lihua, Huang Jingwen, Ge Jirong. Interaction proteomics of long noncoding RNA uc431+ gene in postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(11): 1641-1646. |
| [15] | Chen Qiang, Zhuo Hongwu, Xia Tian, Ye Zhewei . Toxic effects of different-concentration isoniazid on newborn rat osteoblasts in vitro [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1162-1167. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||